The key functions of Qual@xy SPC
Defect map, Process Steering Matrix, Gage R&R: 3 advanced analysis tools
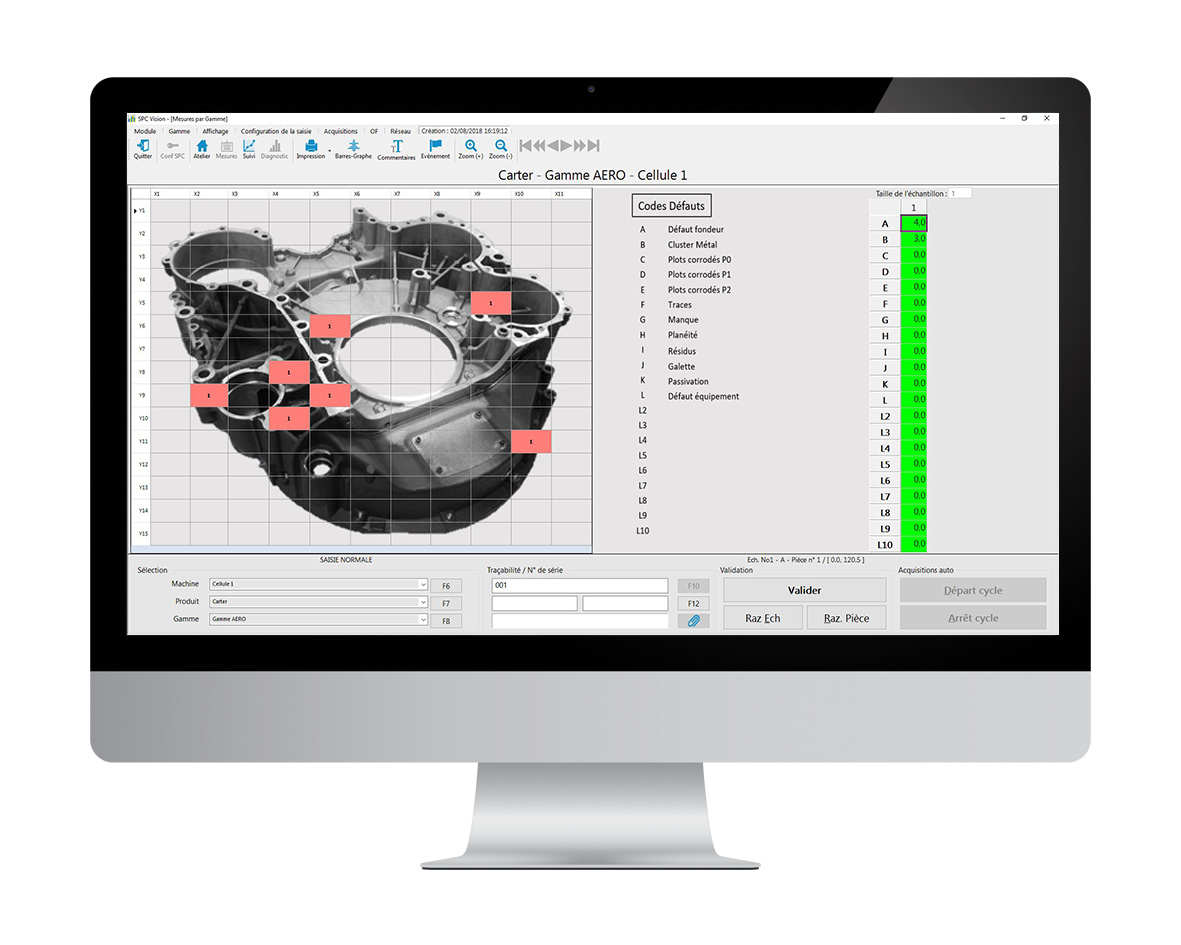
Defect Map
Defect mapping
Defect Map allows you to associate the location and dimensional characteristics of the defects you detect with their location. It is a very powerful tool for analysing all aspects of non-destructive testing in the form of a map: visual, radiology, penetrant testing, ultrasound, etc…
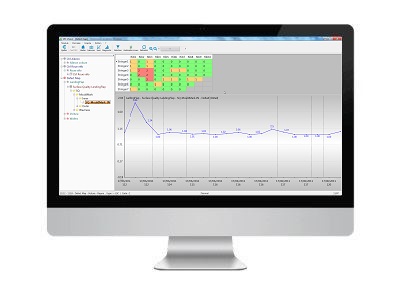
1. Global view of defects
The total number of defects, the general trend and areas of defect occurrence can be tracked.
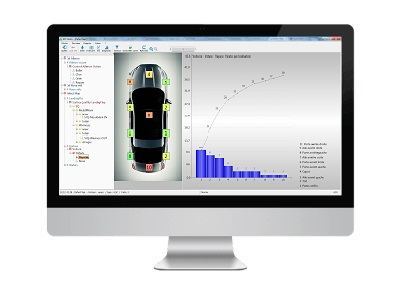
2. Trend for each type of defect
The next step is the trend analysis for each individual defect.
3. Focus your efforts
Give priority to the problems that will generate the greatest improvement. The pareto diagrams show you clearly which defects are to be treated as a priority.
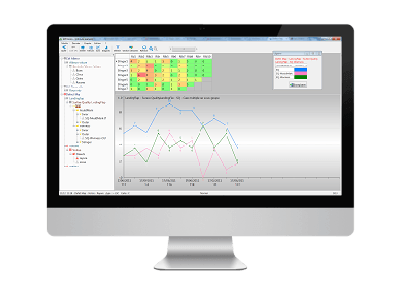
4. Location of the worst defects
Now that you know the extent of the non-conformities and which defect is responsible for the largest number, you can start a root cause analysis.
The Defect Map shows you the areas where defects appear.
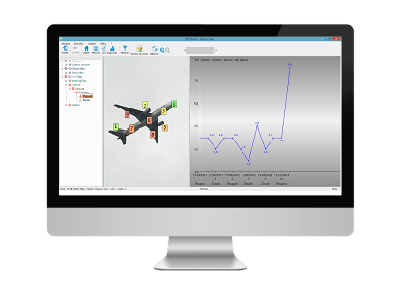
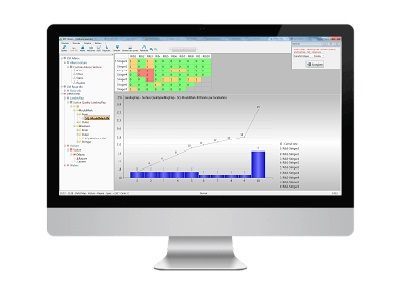
5. In-depth analysis and actions
You now have a clear definition of the problem and a fine exploration that allows you to direct your improvement actions.
You can now tackle the root causes and see the impact of your corrective actions.
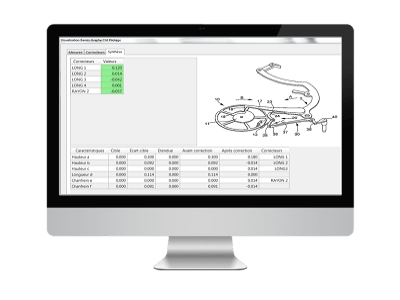
Process Steering Matrix
A control matrix models the links between the machine parameters and the dimensions of the part being manufactured. When the measurements are validated, the control function SPC automatically calculates the best corrections to be made according to the deviations from the target observed on the measured parts.
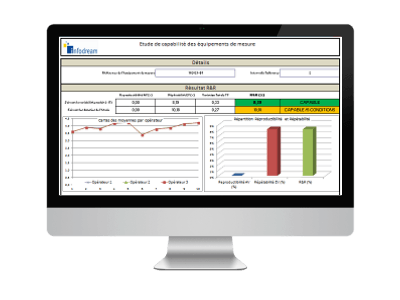
R&R / MSA
Calculation of control process capabilities
The R&R module of the SPC function allows to calculate all the indicators of repeatability and reproducibility required by MSA and other standards, as well as to graphically represent the variation shares. This validation R&R must be a prerequisite for any study SPC.
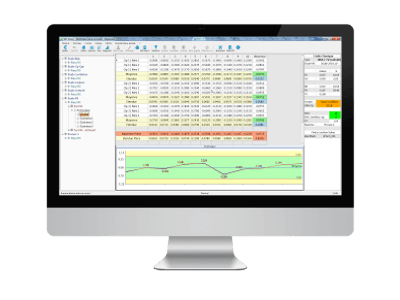
1. Calculation of R&R indicators per study
All measurements are capitalised in the control cards, file by file, with a global vision or a vision by operator or by equipment.
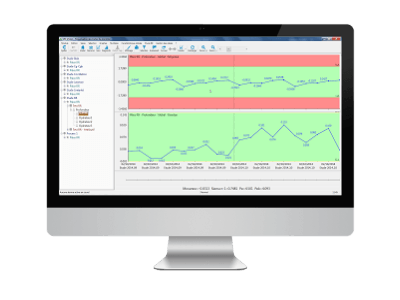
2. Monitoring of indicators over time
The same R&R study can be repeated over time (once a year for example). Qualaxy SPC makes it possible to monitor the evolution of indicators in addition to the current acceptable or unacceptable status.
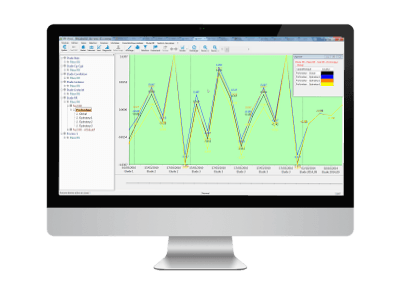
3. Wide range of studies available
R&R control ranges can be configured for different types of study
- classic
- Cg/Cgk/biais
- linearity
- accuracy
- correlation
- repeatability
- attribute
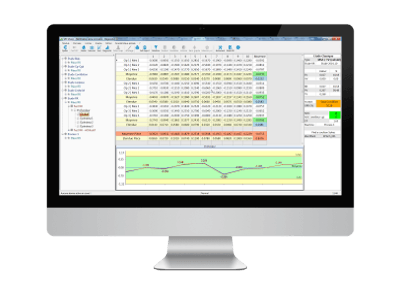
4. Complete, clear and customisable reporting, per study
The format of the « Control Report » file is fully customizable.
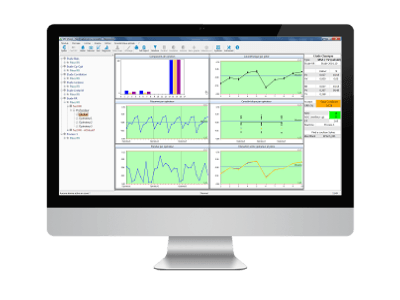
5. A single database
The database is common between SPC and R&R, including for structure objects: a product reference can be followed by a production run and an R&R inspection run. The production data can be linked to the R&R data via the identifiers of the measuring instruments.
Real-time network operation
All data is recorded, analysed and displayed (or edited) in real time. This is absolutely essential for good decision-making.
Main advantages
No more need for off-line analysis
In some companies, we often find engineers who analyse historical data to detect trends, calculate capabilities, etc. These time-consuming activities tell you what the process was at a given moment in the past but do not tell you how your process is behaving at this very moment. Qual@xy SPC presents all data up to date and in real time..
Provoked actions
Qual@xy SPC can be configured to generate predetermined actions under certain conditions.
Accelerating the search for root causes
One of the first tasks in a problem-solving approach is data collection. Very often this data is either non-existent or in a form that requires analysis. This takes time and delays the moment when the root cause can be identified. During this time, the same problem may reappear.
With our Qual@xy SPC module, all data is online, accessible with one or two clicks maximum, whether it is control charts, logs (alarms, events) or capability results.
The process can be managed remotely and in real time.
As Qual@xy SPC is a corporate network system, the data is accessible to everyone, from any workstation. Emails and SMS can even be automatically sent on certain types of incidents. This allows remote monitoring and control of the process with the possibility of analysis in the field.
The plus
All quality analyses and process data are up to date and available to all stakeholders.
SPC & Statistical Analysis
The Qual@xy SPC (formerly SPC Vision) functionality was designed as a quality supervisor. A synoptic of the workshop presents in real time all the current statistical alarms. This way, the user can concentrate only on incidents, without wasting time.
Main advantages
Real-time management of SPC alarms
Non-conformities, drifts or excessive dispersions are detected and highlighted on the synoptic. Corrective actions can be decided upon before a non-conformity occurs or more rejects appear.
Preventive actions are facilitated
One of the objectives of the SPC is to avoid non-quality and associated costs. The early detection of deviations makes this possible. Their qualifications through the intelligence of assignable causes and corrective actions allows a better knowledge of the process. In this way, the opportunity for preventive actions can be highlighted, and can even lead to the planning of maintenance actions which are usually carried out in an emergency.
Permanent measurement of process performance
Very often, companies measure the performance of their process with the rate of non-conformities. This is an a posteriori measurement and few companies really know the ability of their process and machines to meet customer specifications. Qual@xy SPC offers you all the means to compare machines and processes with each other. Thanks to this tool, you can identify the machine/product couples that need improvement or which machines are the most suitable for the manufacture of a given product.
Very rich analysis possibilities
Qual@xy SPC builds all the classic control cards but also allows comparative visualisations, correlations, filtered graphs on certain parameters… In a short time, your knowledge of the process is refined and you discover unsuspected influential factors.
The plus
All available graphical tools and statistical indicators, including :
- Machine and process capabilities according to different standards
- Control charts: mean, range, standard deviation, CUSUM, EWMA… configurable sample size
- Laws: normal, formal defect, unilateral, Rayleigh
- Tests of normality: right of Henry, Chi², Kolmogorov, Anderson-Darling
- Characteristics with attributes: p, np, c, u cards, management of defect typologies and weightings
- Correlation module
- Complete multi-cavity management
- Short series cards
- pareto analysis of events, causes and actions
- Supported standards: Ford « classic », QS9000, ISO TS, AFNOR, CNOMO, user standard on request
- Qual@xy SPC complies with the EN9100 standard
- Qual@xy SPC is FDA 21 CFR Part 11 compliant
Robust and comfortable operator interface
A major asset of Qual@xy SPC is its workshop ergonomics. Operators immediately adopt it for its speed of implementation, comfort and simplicity.
Main advantages
- Operator instructions can be directly integrated into the input interface as text and/or images. This documentation of the inspection operation is dynamic and changes according to the characteristic to be measured
- Qual@xy SPC makes testing simpler and more reliable, saving testing time and error handling costs
- A large number of operations and calculations can be automated
- Validated quality documentation published on a server can be used directly as a support for the capture of measurements: no duplication, assurance of using the right version of the document.
The plus
All available graphical tools and statistical indicators, including :
- The architecture of Qual@xy’s SPC function simulates that of the workshop: operators can easily find their way around it
- Possible formats for the operating mode: rtf, jpg, gif, pdf, and any CAD file
- Various automations: input/output management, sequence of ranges, automatic management of picking…
Measurement management for the company
Qual@xy SPC (the SPC function of the Qual@xy Suite) is a real-time network system that operates a central database.
Main advantages
All data is stored in a single space
- Very often, companies have their production and measurement data stored on different systems, scattered over the network. This situation is limiting because users only use their own data, without a global vision. To be truly profitable, the data must be shared by the widest possible audience in the company, which can then carry out analyses of the entire process and reap the full benefits.
- Qual@xy SPC operates a central database in which data from all workshops, production sites, etc. is recorded, including suppliers if required. In this way, information and communication is fully shared.
Controlled and secure access
All access to the database is secured by a login + password, each user has the rights given to him/her by his/her profile.
The plus
Real-time network operation with automatic outage management
Supported databases: Oracle, SQL Server
Conformity to standards and Traceability
Compliance with standards and traceability management are extremely important for the majority of our customers: this is one of Qual@xy SPC’s strong points. We work intensively with sectors such as aeronautics and pharmaceuticals where standards and regulations are very strict.
Main advantages
Compliance with statistical standards
Qual@xy SPC naturally complies with the main statistical standards:
- FORD, QS9000, ISO TS, AFNOR, CNOMO etc…
- Possibility of customising the standards to adapt to the client’s vocabulary and culture.
Security and version control
One of the objectives of Qual@xy SPC is to avoid non-quality and associated costs. The early detection of deviations makes this possible. Their qualifications through the intelligence of assignable causes and corrective actions allows a better knowledge of the process. In this way, the opportunity for preventive actions can be highlighted, and can even lead to the planning of maintenance actions which are usually carried out in an emergency.
Total traceability and instant search
All measurement or traceability data can be retrieved quickly and easily with Qual@xy SPC. Multi-level traceability such as product structures, serial numbers or batch numbers of OFs etc. allow powerful data management, which allows the complex assembly to be linked to its sub-assemblies, down to the elementary parts.
For example :
Let’s imagine a quality problem during a final assembly phase. With Qual@xy SPC, our SPC module, you immediately find the values obtained on the elementary parts at a stage far removed from the manufacturing process. Conversely, a batch of raw material is defective? Immediately find the list of impacted final assemblies.
These advanced functions incredibly reduce analysis time and facilitate crisis management.
FDA 21 CFR Part 11 compliance
- To the complete satisfaction of our customers in the medical, pharmaceutical and food industries, Qual@xy SPC is FDA 21 CFR Part 11 compliant. This conformity has been certified by the independent and specialist firm: Cyber Conseil.
- Electronic signature.
- Audit trail.
- Non-falsification of data
Integration into the corporate environment
Qual@xy SPC was developed to collaborate with your information system – not duplicate it!
Main advantages
Integration with ERP systems such as SAP
Qual@xy SPC can be fully integrated with your ERP system. Many of our customers launch Qual@xy SPC via their factory ERP. This link can be strengthened: Qual@xy SPC can automatically validate an OF if the measurements are compliant or trigger a non-conformance handling process in case of out-of-tolerance values.
Traceability data, such as OF number, serial number, MSN, etc. are automatically transferred from the ERP system to Qual@xy SPC. The operator does not have to make any double entries. These data are then associated with the measurements and measurements on the product or process. This creates a complete and controllable link between the factory ERP system and the product/process validation data.
Unique architecture for the structure of products and assemblies
Our SPC software has a unique architecture that clearly represents a product and a manufacturing process. The configuration of Qual@xy SPC involves the construction of bricks that are finally assembled to form a ‘production’. This structure allows for very flexible configurations and powerful analyses.
For the production of complex products with high added value and assembly activities (such as in aeronautics), Qual@xy SPC enables a complete structure to be defined. This very powerful structure allows the analysis of a tree structure from the final assembly to the elementary part.
Link Document – Diagram, Instructions, etc.
While the measurements are being recorded, Qual@xy SPC provides the operator with clear control instructions in a user-friendly presentation.
In addition, companies that already have standard documents and control procedures can reuse them directly under Qual@xy SPC: simply create links to these documents, no duplication is necessary. The version of the document is controlled… the paperless workshop is there!
Integration of the Non-Compliance Management System
Usually the management of non-conformities is a function of ERP. Qual@xy SPC does not duplicate but enhances this function: Non-conformity management ERPs often generate a delay between the detection of a non-conformity and the decision to take corrective action due to a lack of detail on the nature of the non-conformity. Qual@xy SPC software can significantly reduce this delay by providing the decision-maker with data in real time and in a clear and concise format.
Support for quality improvement programmes such as 6 Sigma, etc.
Qual@xy SPC has been developed in a spirit of continuous improvement. The analysis capabilities and reporting functions are directly linked to programmes such as 6 sigma.